Effects of Upper-Extremity Heat Stress Combined with Resistance Training on Skill and Basal Fitness, Isokinetic Muscle Strength, and Anabolic Hormones in Wheelchair Rugby Athletes with Spinal Cord Injury
Article information
Abstract
Background
This study aimed to investigate the effects of upper-extremity heat stress combined with resistance training and resistance training alone on body composition, skill and basal fitness, isokinetic muscle strength, and anabolic hormone levels in wheelchair rugby athletes with spinal cord injury.
Methods
Fourteen male national wheelchair rugby athletes with cervical-level spinal cord injury were randomly assigned to the heat stress combined with resistance training (n=7) or resistance training group (n=7). Both groups completed a 10-week upper-extremity resistance training program. Additionally, heat stress was applied to the upper-extremity of the heat stress combined with resistance training group using steam-generating thermal sheets three times per week. Measurements conducted pre- and post-intervention included body composition, skill and basal fitness (one-hand throw, chest throw, 28 m shuttle sprint, and 20 m sprint), isokinetic strength (using the Cybex system at 60°/sec and 180°/sec), and anabolic hormones (growth hormone and insulin-like growth factor). Non-parametric tests (Wilcoxon signed-rank and Mann-Whitney U test) were used for statistical analysis (α=0.05).
Results
The heat stress combined with resistance training group showed significant increases in skeletal muscle mass and upper-extremity isokinetic elbow joint strength, particularly in flexion (%BW) and extension (%BW) at 60°/sec. Growth hormone levels increased significantly only in this group, while insulin-like growth factor levels showed marginal improvement. Both groups improved in 28 m shuttle sprint and one-hand throw; however, the heat stress combined group showed a greater effect size in the 20 m sprint. No significant changes were observed in body mass index or body fat percentage.
Conclusions
The combination of heat stress and resistance training may provide additional benefits in enhancing upper-extremity muscle strength and stimulating anabolic hormone responses in wheelchair rugby athletes with spinal cord injury. These findings suggest a promising and practical strategy for optimizing training adaptations in this athletic population.
INTRODUCTION
Spinal cord injury (SCI) refers to the partial or complete impairment of the spinal cord resulting from trauma or disease. Such injuries induce a wide range of degenerative changes below the level of the lesion, particularly affecting musculoskeletal and metabolic functions [1,2]. Muscle atrophy following SCI is characterized by a marked reduction in slow-twitch muscle fibers and a phenotypic shift toward fast-twitch fiber predominance, which leads to decreased oxidative capacity and accelerated fatigue [3,4]. Concurrently, the lack of mechanical loading promotes bone resorption and results in a rapid decline in bone mineral density [5]. These physiological alterations, along with ectopic fat accumulation in atrophied muscles, contribute to reduced insulin sensitivity and increase the risk of type 2 diabetes and cardiovascular complications [6]. This in turn negatively affects independence in daily activities and overall quality of life [7,8].
Rehabilitation exercises have been emphasized as an intervention strategy to compensate for these functional impairments. Numerous studies have demonstrated that resistance training (RT) effectively maintains or increases muscle mass, improves body composition, and restores muscle function [9,10]. However, in individuals with SCI, the application of training programs requires careful consideration because the upper-extremity serves as the primary means of mobility and performance of activities of daily living. The upper-extremity serves as the primary means of wheelchair propulsion and weight-bearing activities, and more than 70% of individuals with SCI experience upper-extremity pain [11,12].
These characteristics are particularly evident in wheelchair sports. Wheelchair rugby is a high-intensity sport that requires substantial upper-extremity strength, muscular endurance, and technical proficiency due to the physical demands of the game [13]. Athletes engage in repetitive rotations, directional changes, acceleration, and sprinting motions, making the enhancement of upper-extremity function a crucial factor not only for sports performance but also for independent activities [14]. However, repetitive wheelchair propulsion and intense physical contact during competition expose the upper-extremity muscles and shoulder joint to continuous mechanical loading, which may increase the risk of chronic overuse injuries [15]. Consequently, high-intensity RT alone may place excessive stress on the shoulder joint, potentially increasing the risk of musculoskeletal injury. Considering these limitations, adjunctive intervention strategies are needed to alleviate muscle fatigue and tissue load while maintaining the benefits of RT. Recent studies have focused on the combination of rehabilitation exercise with adjunctive methods, such as heat stress, with a view to enhancing therapeutic effectiveness [16,17].
Heat stress induces various physiological responses, including increased local tissue temperature and blood flow, stimulation of protein synthesis, removal of metabolic waste products, reduction of inflammation, and stimulation of growth hormone (GH) and insulin-like growth factor-1 (IGF-1) secretion [18,19]. Animal and cellular studies show that repeated heat exposure promotes muscle hypertrophy and strength by enhancing protein synthesis, reducing atrophy, and activating satellite cells. Heat-induced proteins such as HSP72 facilitate protein repair and cellular protection, while increased blood flow through vasodilation improves metabolic waste removal and oxygen and nutrient delivery, supporting recovery and microvascular function [20]. Heat stress improves muscle oxygen delivery and reduces muscle fatigue before and after exercise, showing potential as a means of enhancing exercise effectiveness in athletes performing high-intensity exercises or in patients with functional limitations [21].
In particular, the application of heat stress before and after RT may promote peripheral vasodilation and improve soft-tissue flexibility, thereby reducing injury risk, enhancing training safety, and potentially augmenting the physiological benefits of RT [22]. However, research analyzing the effects of combined heat stress and RT interventions on body composition, skill and basal fitness, muscular function, and endocrine responses in individuals with SCI disabilities, particularly wheelchair athletes, is limited. Therefore, this study aimed to analyze changes in body composition, skill and basal fitness, isokinetic strength, and anabolic hormones induced by a 10-week intervention combining heat stress and RT (heat with resistance training, HRT) in the upper-extremity of wheelchair rugby athletes with SCI compared to RT alone.
METHODS
Participants
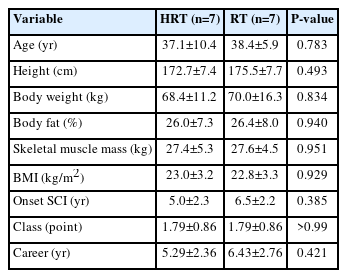
Twenty male national wheelchair rugby athletes from the Korea Wheelchair Rugby Association provided informed consent and were recruited. After excluding individuals with cardiovascular conditions or medication risks, 14 were finally selected to participate in the study. A priori power analysis indicated a required sample size of 20 participants (effect size [ES] f=0.35, α=0.05, power=0.80; G*Power 3.1.9.7), but attrition reduced the final sample to 14. Participants had cervical spinal cord injuries (C4–C7) or spinal muscular atrophy and were randomly assigned to the HRT or RT groups based on wheelchair rugby classification. Analysis of pre-training body composition for both the HRT and RT groups is presented in Table 1. No significant differences were observed between the groups in terms of age, height, weight, body fat percentage (BF%), skeletal muscle mass (SMM), body mass index (BMI), or SCI onset.
Study design
Experimental procedure
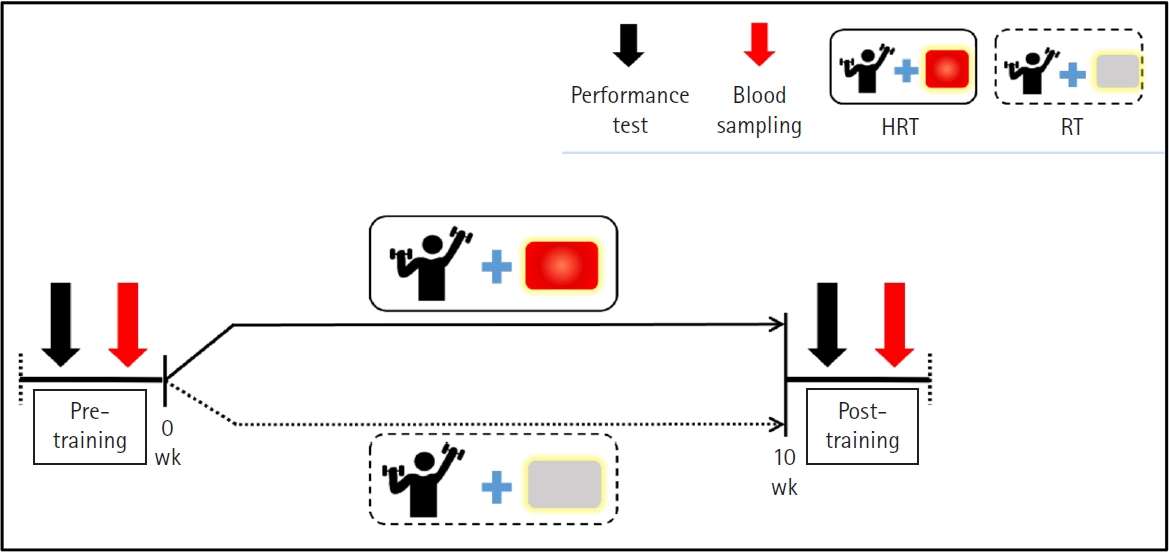
All participants were informed of the study purpose and procedures and were randomly assigned to the HRT or RT groups. The HRT group received combined heat application and RT, while the control group received placebo treatment with RT. Both groups completed a 10-week program, with performance tests and blood sampling conducted pre- and post-training the intervention. The study design is presented in Fig. 1. This study was approved by the Korea University Institutional Review Board (No. KUIRB-2024-0114-01) and conducted in accordance with the Declaration of Helsinki.
Resistance training
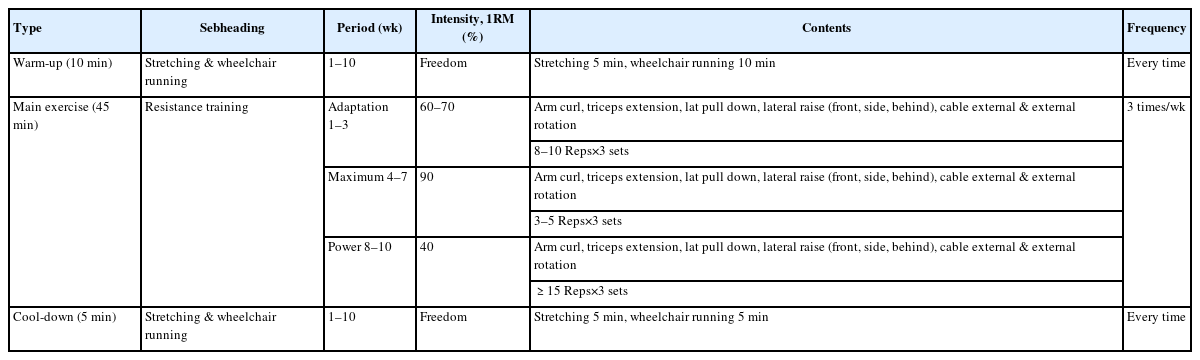
All participants engaged in a 10-week residential training program that incorporated a structured RT component using standard weight-training equipment. The one-repetition maximum for each participant was estimated using the Smith machine bench press protocol based on the indirect prediction equation proposed by Mayhew et al. [23]. The training program was divided into three progressive phases: weeks 1–3, which focused on anatomical adaptation; weeks 4–7, which emphasized maximal strength development; and weeks 8–10, which targeted power improvement. The exercise intervention consisted of a 10-minute warm-up, 45 minutes of main exercise, and a 5-minute cool-down. The detailed training parameters for each phase are presented in Table 2.
Heat application
Heat- and steam-generating sheets (Kao Corporation) were applied to the biceps and triceps of both arms to increase muscle temperature. The safety of these sheets for human use has been verified by the Kao Corporation Safety Committee [24]. In this study, the heat sheets were applied exclusively to participants in the HRT group. The sheets were applied 6–8 hours before the start of the exercise session. Participants performed the exercise while wearing the heat sheets, which were removed after the completion of the exercise. The sheets were attached for 6–8 hours per day, three times per week. This application duration was determined based on previous studies reporting that thermal stimulation may influence biochemical and physiological changes in skeletal muscle [25,26]. In contrast, participants in the RT group used identical sheets with deactivated heat-generating functions. The application schedule and duration were matched with those of the HRT group to ensure experimental consistency.
Measurements and methods
Body composition
Body composition was assessed using a bedside bioelectrical impedance analyzer (InBody S10; Biospace). Participants rested supine for 10 minutes before measurement to ensure stabilization. During testing, they maintained a straight posture with arms extended approximately 15° from the torso and legs shoulder-width apart to prevent contact. Electrodes were attached to the thumbs and middle fingers and placed between the medial malleolus and heel on both sides. Participants remained still throughout the procedure. Measured variables included BF%, fat mass, SMM, and BMI for subsequent analysis.
Skill and basal fitness
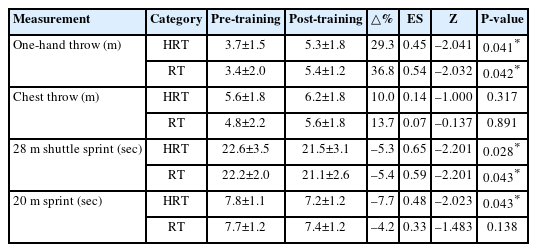
We used modified version of the Beck Battery of Quad Rugby Skill Tests developed by Yilla and Sherrill [27] and De Groot et al. [28] was used to assess skill and basal fitness in wheelchair rugby athletes. The test included three components: (1) Explosive power and accuracy – Distance and accuracy throws using an official wheelchair rugby ball. One-hand throw and chest throw were performed three times each, and the mean value was analyzed. (2) Cardiorespiratory endurance – A 28 m shuttle sprint (out and back), with the average time of three trials recorded. (3) Anaerobic power – A 20 m sprint, with the mean time of three trials used for analysis.
Isokinetic muscle strength
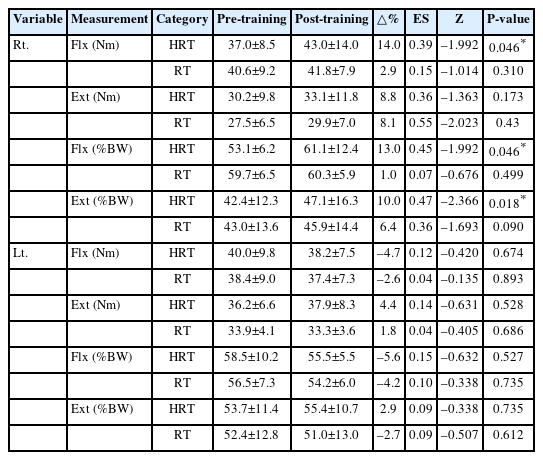
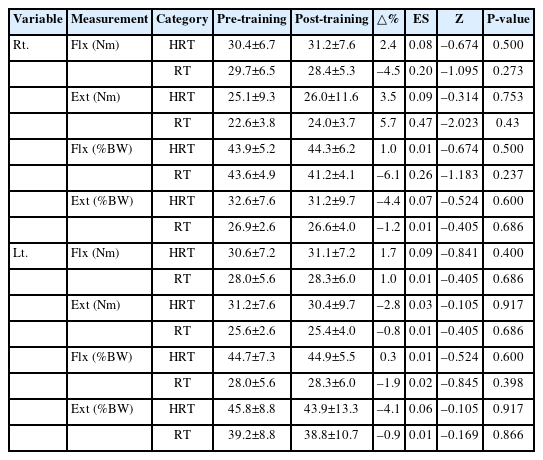
The isokinetic muscle strength of the elbow joints was assessed using an isokinetic dynamometer (Cybex Humac Norm) to measure both peak torque and relative strength. Measurements were conducted with participants placed in the supine position on a Cybex examination chair. The trunk and legs were secured with straps to ensure stability. The arm was positioned close to the torso, and the shoulder was aligned with the dynamometer’s axis of rotation. Elbow joint strength was then assessed in the position, with the elbow joint flexed to 90° and warmed up through 0°–100°. Bilateral elbow flexor and extensor strength were measured at 60°/sec and 180°/sec. The hand was strapped to the grip for consistency, and each muscle group was tested three times to determine peak torque.
Anabolic hormones (growth hormone and insulin-like growth factor-1)
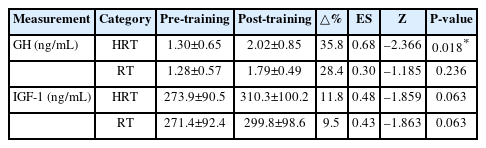
Approximately 7 mL of resting venous blood was drawn from the antecubital vein by a nurse at baseline and after the 10-week intervention. Blood samples were placed in serum separation tubes and gently inverted 5–10 times to mix the clot activator and separation gel, then centrifuged (MF 300) at 3,000 rpm for 15 minutes to separate serum. The serum was stored at –80 °C until analysis. The blood samples were analyzed for the anabolic hormones GH and IGF-1 by a specialized clinical laboratory.
Statistical analysis
All data were analyzed using SPSS software (version 22.0; IBM Co.). Descriptive statistics are presented as mean and standard error of the mean. Normality tests revealed that several variables did not meet the assumption of normal distribution; therefore, non-parametric statistical methods were employed. Between-group differences were analyzed using the Mann-Whitney U test, and within-group (pre–post) comparisons were conducted using the Wilcoxon signed-rank test. All values are reported as percentage change, ES, Z-value, and P-value. The ES was calculated using a rank-biserial correlation, which is appropriate for nonparametric tests. The thresholds for interpreting the ESs were as follows: r=0.1 (small); r=0.3 (moderate); r=0.5 (large). The level of statistical significance was set at P=0.05.
RESULTS
Body composition
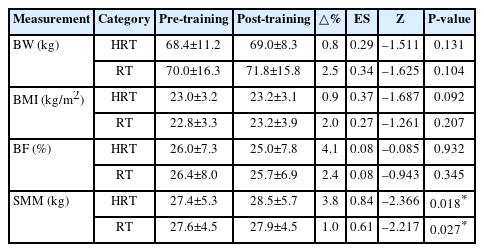
The changes in body composition indicators pre- and post-training for both groups the HRT and RT groups are presented in Table 3. No significant changes were observed in body weight, BMI, or BF% in either group. However, SMM significantly increased from pre- and post-training in both the HRT (Z=–2.366, P=0.018) and RT (Z=–2.217, P=0.027) groups.
Skill and basal fitness
The changes in skill and basal fitness indicators pre- and post-training for both the HRT and RT groups are presented in Table 4. Significant improvements were noted in the one-hand throw in the HRT (Z=–2.041, P=0.041) and RT groups (Z=–2.032, P=0.042) post intervention. However, no significant changes were observed in the chest throw results in either group. The 28 m shuttle sprint significantly decreased in the HRT (Z=–2.201, P=0.028) and RT groups (Z=–2.201, P=0.043) post intervention. Conversely, the 20 m sprint significantly decreased only in the HRT group post intervention (Z=–2.023, P=0.043), with no significant changes observed in the RT group.
Isokinetic strength changes
The changes in isokinetic elbow joint strength indicators, measured at 60°/sec pre- and post-training, for both HRT and RT groups are presented in Fig. 2A, B and Table 5. Right elbow joint flexion strength (Nm) significantly increased in the HRT group post training (Z=–1.992, P=0.046), however, no significant changes were observed in the RT group. No significant changes were observed in right elbow joint extension strength (Nm) in either group post-training (Fig. 2A). Relative right elbow joint flexion and extension strength relative to body weight (percentage of body weight, %BW) significantly increased post-training only in the HRT group (Z=–1.992, P=0.046; Z=–2.366, P=0.018), whereas no significant changes were observed in the RT group. The left elbow joint flexion and extension strength (Nm) and relative left elbow joint flexion and extension strength relative to body weight (%BW) showed no significant changes in either group (Fig. 2B).
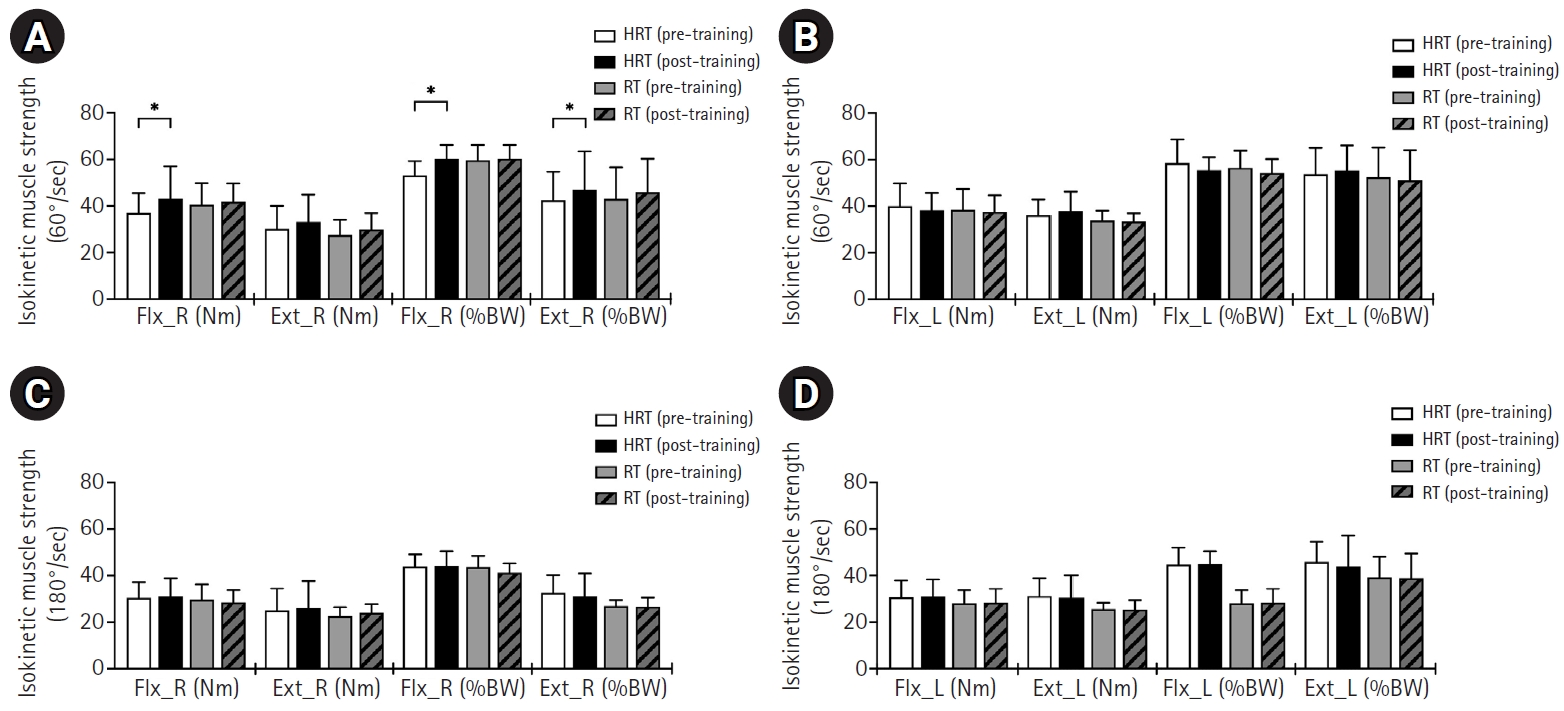
Changes in isokinetic elbow joint strength pre- and post-training in HRT and RT groups. Wilcoxon signed-rank and Mann-Whitney U tests were used. (A) Right elbow joint flexion and extension strength at 60°/sec. (B) Left elbow joint flexion and extension strength at 60°/sec. (C) Right elbow joint flexion and extension strength at 180°/sec. (D) Left elbow joint flexion and extension strength at 180°/sec. Ext_L, left elbow joint extension; Ext_R, right elbow joint extension; Flx_L, left elbow joint flexion; Flx_R, right elbow joint flexion; HRT, heat with resistance training; RT, resistance training; %BW, percentage of body weight (strength relative to body weight). *Significant difference between pre- and post-training in the HRT group (P<0.05).
The changes in isokinetic elbow joint strength indicators, measured at 180°/sec pre- and post-training, for both HRT and RT groups are presented in Fig. 2C, D and Table 6. No significant changes were observed in the right or left elbow joint flexion and extension strength (Nm) or relative flexion and extension strength relative to body weight (%BW) in either group.
Hormonal changes
The changes in anabolic hormones analysis indicators pre- and post-training for both the HRT and RT groups are presented in Table 7. GH significantly increased post-training only in the HRT group (Z=–2.366, P=0.018), whereas no significant change was observed in the RT group. No significant changes were observed in IGF-1 levels in either group.
DISCUSSION
The present study investigated the effects of a 10-week intervention combining upper-extremity HRT on body composition, skill and basal fitness, isokinetic muscle strength, and anabolic hormone responses in wheelchair rugby athletes with SCI. Although no significant inter-group differences were observed, the HRT intervention enhanced isokinetic strength, GH levels, and 20 m sprint performance compared with RT alone. These findings suggest that the combination of localized heat stress and resistance exercise may contribute to improvements in neuromuscular function in athletes with SCI.
In the present study, both the HRT and RT groups demonstrated significant increases in SMM, whereas body weight, BMI, and BF% remained unchanged. This pattern suggests that the intervention selectively promoted lean tissue accretion without affecting overall body mass or adiposity. The increase in muscle mass in both groups is consistent with previous findings that RT, even over relatively short intervention periods, can enhance muscle hypertrophy in individuals with SCI [29,30]. The lack of change in total body weight or body fat may be attributed to the small energy expenditure relative to total daily energy balance, or the high baseline training status of the participants, which may limit fat mass reductions [31].
The results of the present study showed that both the HRT and RT groups improved in the one-hand throw, whereas no significant changes were observed in the chest throw test. The improvement in one-hand throw performance may be related to enhanced force generation during unilateral upper-extremity movements [32].
The one-hand throw primarily relies on rapid elbow extension and flexion to accelerate the ball, and repeated propulsion RT may improve the ability to produce force during the acceleration phase of the throwing motion [33]. As a result, athletes may have been able to generate greater throwing velocity and distance. In contrast, the chest throw requires bilateral and coordinated force production with greater trunk involvement [34]. Although RT may improve upper-extremity strength, trunk muscle activation and stabilization capacity can remain limited in individuals with SCI due to impaired neural control [35]. As a result, improvements in upper-extremity strength may not necessarily translate into enhanced performance in tasks that require substantial trunk involvement. This is supported by the absence of improvement in the chest throw therefore suggests that the training adaptations were movement and limb specific, consistent with the principle of training specificity [36,37].
Regarding sprint performance, both the HRT and RT groups improved in the 28 m shuttle sprint, whereas only the HRT group improved in the 20 m sprint. The greater improvement observed in the HRT group in the 20 m sprint may reflect the additional effects of localized heat stress, which has been reported to increase blood flow, reduce muscle fatigue, and improve neuromuscular efficiency during high-intensity exercise [38,39]. In contrast, the 28 m shuttle sprint involves repeated directional changes and a slightly longer movement duration, which may rely more heavily on overall wheelchair propulsion capacity and repeated upper-extremity force production developed through RT [40,41]. Consequently, improvements in shuttle sprint performance were observed in both groups, whereas the shorter, high-intensity 20 m sprint appeared to be more responsive to the additional effects of heat stress. The present findings indicate that combining localized HRT may enhance neuromuscular adaptations in athletes with SCI.
Although no significant inter-group differences were observed, the HRT group demonstrated selective improvements in right elbow flexion peak torque and %BW, as well as right elbow extension %BW, highlighting potential enhancements in upper-extremity force production relevant to wheelchair propulsion. These selective adaptations likely reflect the functional demands of wheelchair rugby, where propulsion and ball-handling often involve asymmetric, dominant limb dependent force production [42,43]. Elbow flexors contribute during the recovery phase of propulsion, facilitating efficient arm repositioning, while elbow extensors generate propulsive force during the push phase [44]. The improvements in these specific joint actions suggest enhanced neuromuscular efficiency and motor unit recruitment in the dominant limb, supporting faster acceleration and improved sprint performance [45]. The lack of changes in other isokinetic measures may be attributed to the short duration of the intervention or the already high baseline upper-body strength of trained wheelchair rugby athletes. In highly trained individuals, adaptations are often task-specific, occurring primarily in muscles and movement patterns most frequently engaged during sport-specific activity [36]. Overall, these results suggest that HRT can selectively augment upper-extremity strength in movement patterns critical for wheelchair propulsion, potentially enhancing performance in sprint and maneuvering tasks that are central to wheelchair rugby.
In the present study, a significant increase in GH levels was observed only in the HRT group, whereas IGF-1 did not show significant changes in either group. GH is known to play an important role in protein synthesis, muscle hypertrophy, and tissue repair following exercise [46]. Previous studies have presented that RT can stimulate GH secretion, particularly when exercise is performed at relatively high intensities or with large muscle mass involvement [47]. In the present study, the additional increase in GH observed in the HRT group may be associated with the physiological stress induced by localized heat exposure. Heat stress has been reported to stimulate endocrine responses, including increased GH secretion, possibly through thermoregulatory stress and elevated metabolic demand [48]. In contrast, IGF-1 levels did not significantly change in either group. Circulating IGF-1 responses are known to be variable and influenced by multiple systemic factors, including nutritional status, liver function, and overall metabolic regulation [49]. Another contributing factor may be that the participants were trained wheelchair rugby athletes, in whom hormonal adaptations to RT are often attenuated due to prior physiological adaptation [47]. Finally, muscle adaptation can occur through local IGF-1 expression within skeletal muscle without altering circulating concentrations, indicating that the lack of significant change in serum IGF-1 does not necessarily reflect the absence of anabolic adaptations.
To our knowledge, this is the first study to investigate combined heat stress and upper-extremity RT in wheelchair rugby athletes with SCI. Although the effects were modest and condition-dependent, the results suggest that the unique physiological characteristics of athletes with disabilities may enhance the benefits of heat stress, supporting its integration into rehabilitation training. Future research should include longer interventions, athletes from various wheelchair sports, and additional training stimuli such as heat exposure and blood flow restriction tailored to adaptive sports populations. One limitation of this study is that the final sample size was slightly smaller than the number suggested by the a priori power analysis, which may have reduced the statistical power of the study. Therefore, the findings should be interpreted as preliminary evidence, and future studies with larger sample sizes are warranted.
In conclusion, although no significant inter-group differences were observed, HRT showed potential for enhancing muscular strength and function in athletes with SCI. The combination of upper-extremity heat stress and RT improved isokinetic strength, GH levels, and 20 m sprint performance compared to RT alone, suggesting enhanced neuromuscular function. No significant changes were observed in body fat or BMI. These findings indicate that localized heat stress may be an effective adjunct to RT, warranting further research to refine protocols and examine long-term outcomes.
Notes
AUTHOR CONTRIBUTIONS
Dr. Sung Jin YOON had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. All authors reviewed this manuscript and agreed to individual contributions.
Conceptualization: MJL and JML. Data curation: MJL and JML. Formal analysis: SJY. Methodology: MJL and JML. Writing–original draft: MJL and JML. Writing–review & editing: MJL and SJY.
CONFLICTS OF INTEREST
No existing or potential conflict of interest relevant to this article was reported.
FUNDING
None.
DATA AVAILABILITY
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.